—
Boltzmann plot of the first electronic excited states of carbon.#
The equilibrium is computed via Cantera at a given temperature and pressure.
From Boltzmann equation, we have:
\[n_\text{u} = n_0 \frac{g_\text{u} \exp\left(-\frac{E_\text{u}}{k_\text{B} T}\right)}{Z(T)}\]
Dividing by the total density, and rearranging terms, we obtain:
\[\log\left(\frac{x_\text{u}}{g_\text{u}}\right) =
\log\left(\frac{x_\text{0}}{Z(T)}\right)
-\frac{E_\text{u}}{k_\text{B} T}\]
# This is an option for the online documentation, so that each image is displayed separately.
# sphinx_gallery_multi_image = "single"
Import the required libraries.#
import cantera as ct
import matplotlib.pyplot as plt
import numpy as np
from adjustText import adjust_text
import rizer.misc.units as u
from rizer.kin.extensible_rate import * # noqa: F403
from rizer.misc.plt_utils import set_mpl_style
from rizer.misc.utils import get_path_to_data
set_mpl_style(nb_columns=1)
Define mechanisms and parameters.#
mechanism = get_path_to_data(
"mechanisms",
"Goutier2025",
"CH4_to_C2H2.yaml",
)
gas = ct.Solution(mechanism, name="gas_with_electronic_excited_states")
assert "C(1D)" in gas.species_names
# Temperatures in Kelvin.
temperatures = np.array([2000, 5000, 10000, 15000, 20000, 30000, 40000, 50000])
# Pressure in Pascals.
P = ct.one_atm
# Initial mole fraction (here, we start with pure methane).
X_0 = "CH4:1"
# Energy and degenaracy of the first electronic excited states of carbon.
# cf. https://physics.nist.gov/cgi-bin/ASD/energy1.pl?de=0&spectrum=C+I&units=1&format=0&output=0&page_size=15&multiplet_ordered=0&conf_out=on&term_out=on&level_out=on&unc_out=1&j_out=on&g_out=on&lande_out=on&perc_out=on&biblio=on&temp=&submit=Retrieve+Data
energies_eV = np.array([0.0, 1.2637284, 2.6840136, 4.1826219])
gs = np.array([9, 5, 1, 5])
Plot the Boltzmann plot.#
fig, ax = plt.subplots()
texts = []
for T in temperatures:
# Equilibrate the mixture.
gas.TPX = T, P, X_0
gas.equilibrate("TP")
# Get the mole fraction.
X = gas["C", "C(1D)", "C(1S)", "C(5So)"].X
# Plot log(x/g) vs energy
ax.scatter(energies_eV, np.log(X / gs), marker="o", s=300)
# Fit data to `y=b-a*x`
coefs = np.polynomial.polynomial.polyfit(x=energies_eV, y=np.log(X / gs), deg=1)
ax.plot(
energies_eV,
np.polynomial.polynomial.polyval(energies_eV, coefs),
ls="--",
lw=4,
)
# Get back the temperature.
# y = b - a*x
a_per_eV = -coefs[1] # eV^(-1)
a_per_J = a_per_eV / (u.eV_to_J) # J^(-1)
# a = 1/(k_B * T)
T_fit = 1 / (a_per_J * u.k_b) # K
print(f"Temperature from fit: {T_fit:.2e} K")
# Annotate at 2 eV.
texts.append(
ax.text(
x=2,
y=float(np.polynomial.polynomial.polyval(2, coefs)),
s=f"{int(T_fit):,} K".replace(",", " "),
color=ax.lines[-1].get_color(),
horizontalalignment="center",
verticalalignment="center",
bbox=dict(
facecolor="white",
alpha=0.8,
edgecolor=ax.lines[-1].get_color(),
boxstyle="round",
),
)
)
ax.set_xlabel(r"$\varepsilon$ [eV]")
ax.set_ylabel(r"$\log\left(\frac{x}{g}\right)$")
ax.set_title("Boltzmann plot of the first 4 electronic excited states of carbon.")
adjust_text(texts, arrowprops=dict(arrowstyle="->", color="k"))
plt.show()
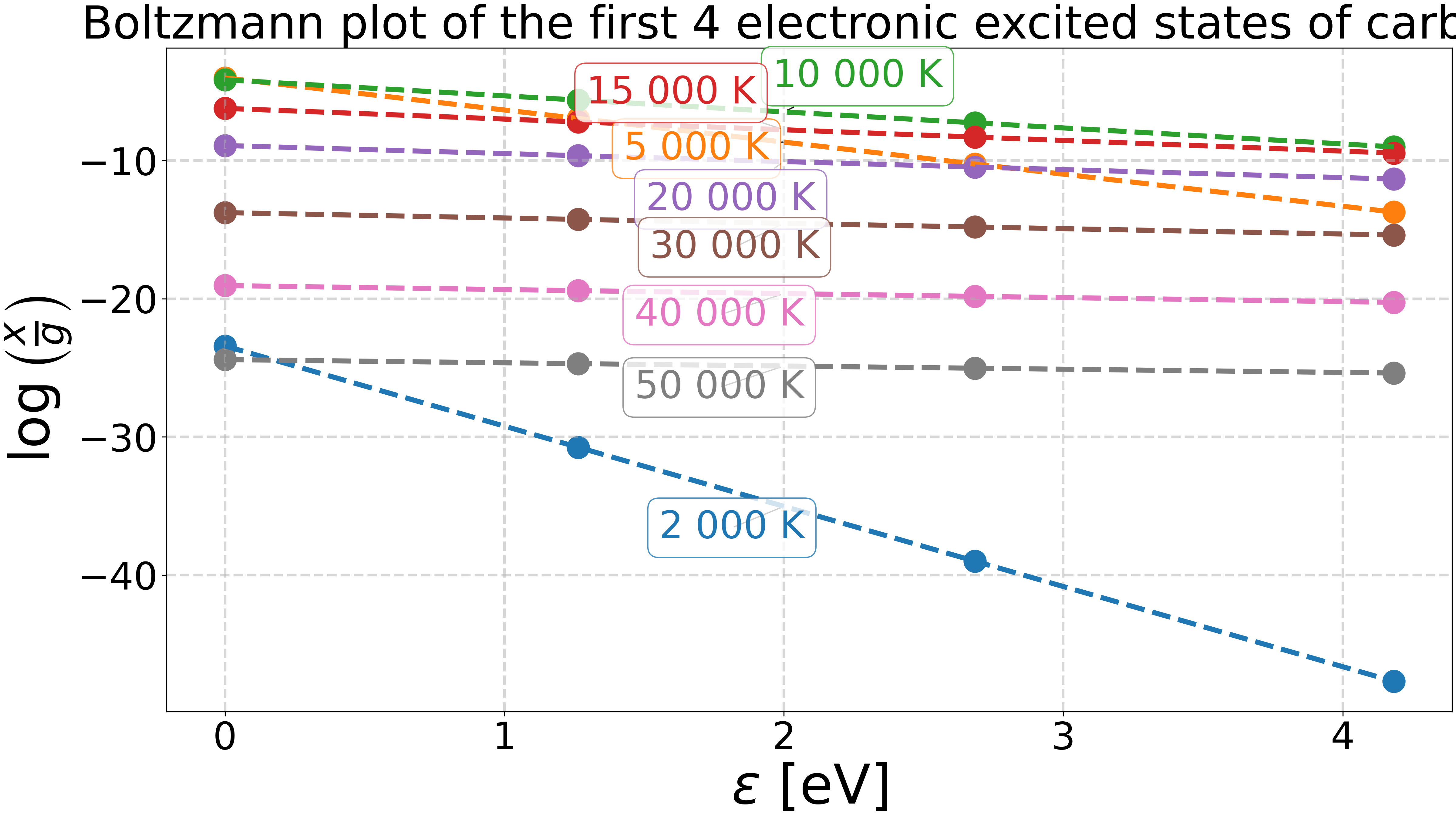
Temperature from fit: 2.00e+03 K
Temperature from fit: 5.00e+03 K
Temperature from fit: 1.00e+04 K
Temperature from fit: 1.50e+04 K
Temperature from fit: 2.00e+04 K
Temperature from fit: 3.00e+04 K
Temperature from fit: 4.00e+04 K
Temperature from fit: 5.00e+04 K
Total running time of the script: (0 minutes 1.597 seconds)
